 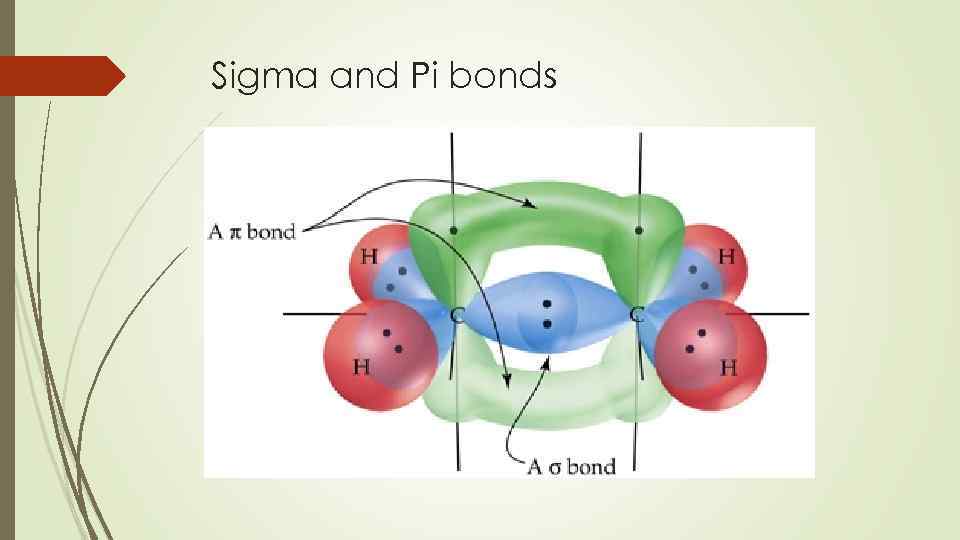
Here, we see another element of stereopecificity: Diels-Alder reactions with cyclic dienes favor the formation of bicyclic structures in which substituents are in the endo position. 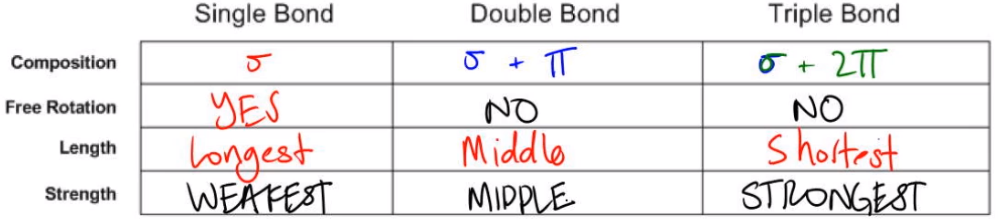
The result of a Diels-Alder reaction involving a cyclic diene is a bicyclic structure: 
Cyclic dienes, on the other hand, are ‘locked’ in the s-cis conformation, and are especially reactive. For some dienes, extreme steric hindrance causes the s-cis conformation to be highly strained, and for this reason such dienes do not readily undergo Diels-Alder reactions. The number of sigma and pi-bonds in 1-butene 3-yne are. A cis dienophile will generate a ring with cis substitution, while a trans dienophile will generate a ring with trans substitution: In order for a Diels-Alder reaction to occur, the diene molecule must adopt what is called the s-cis conformation: The s-cis conformation is higher in energy than the s-trans conformation, due to steric hindrance. Sigma and pi bonds are an aspect of valence bond theory and molecular orbital theory that explains the existence of double and triple bonds. The hybrid state of C-atoms which are attached to a single bond with each other in the following. So there are a total of 12 bonds and 3 bonds in benzene. In bemzene a double bond is present in between alternate carbon atoms and each carbon is attached to two carbon atoms by means of a single bond and a double bond and a hydrogen atom by a single bond. Its 2p z orbital will form a pi bond with the other oxygen’s 2p z orbital. Benzene molecule contains 12 and 3 bonds. Example: In carbon dioxide (CO 2), the 2p x orbital of carbon will form a pi bond with the unhybridized 2p x orbital of oxygen. A triple bond is made up of one sigma and two pi bonds. Both of these new overlaps end up forming new sigma bonds, and a new pi bond is formed between carbon 2 and 3 of the diene.One of the most important things to understand about this process is that it is concerted – all of the electron rearrangement takes place at once, with no carbocation intermediates.The Diels-Alder reaction is enormously useful for synthetic organic chemists, not only because ring-forming reactions are useful in general but also because in many cases two new stereocenters are formed, and the reaction is inherently stereospecific. A double bond consists of one sigma and one pi bond. Essentially, this process involves overlap of the 2p orbitals on carbons 1 and 4 of the diene with 2p orbitals on the two sp 2-hybridized carbons of the dienophile. In a Diels-Alder reaction, the alkene reacting partner is referred to as the dienophile. In the Diels-Alder cycloaddition reaction, a conjugated diene reacts with an alkene to form a ring structure. It explains how to calculate the number of sigma and pi bonds in a molecule given its lewis structure. Ionisation energy order is N2 > O2 Xe > NO. This chemistry video tutorial provides a basic introduction into sigma and pi bonds. We end this chapter with a discussion of a type of reaction that is different from anything we have seen before. B2, C2 are examples of species having no sigma but only pi bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
